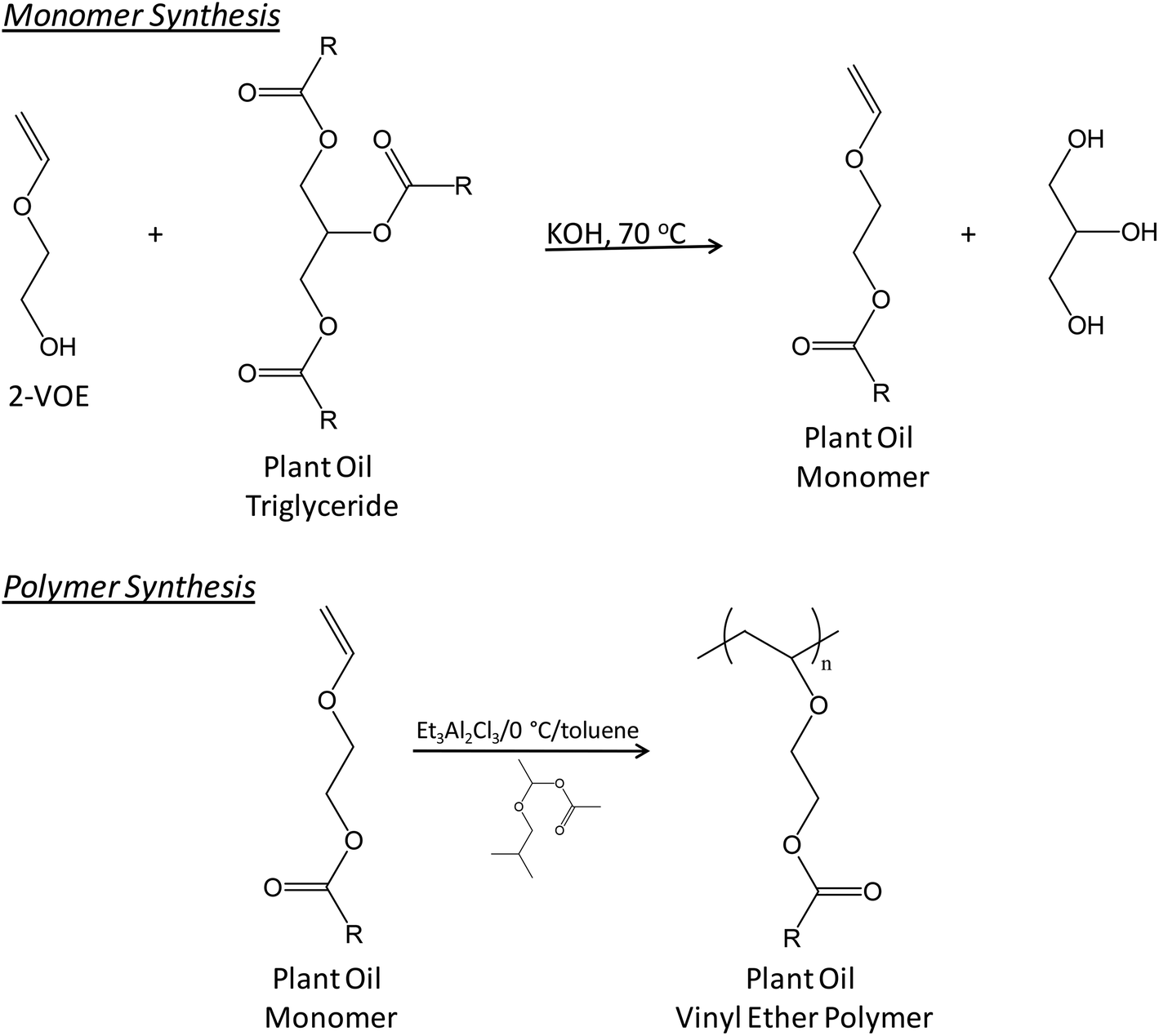
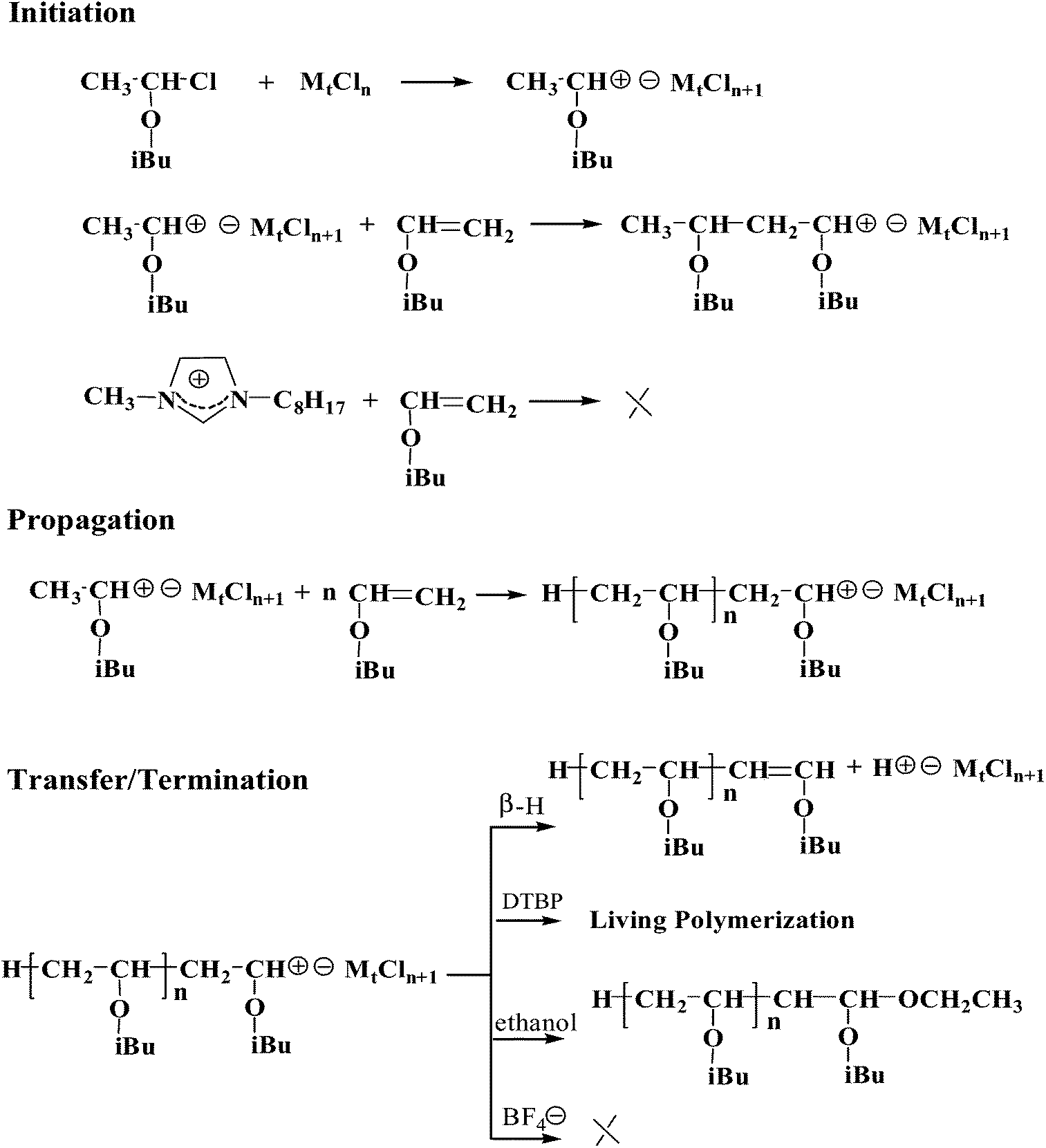The polymerization proceeded in a reproducible manner through the careful design of experimental conditions adding initiator co solvents and.
Vinyl ether polymerization mechanism.
The mechanism of the recently reported photocontrolled cationic polymerization of vinyl ethers was investigated using a variety of catalysts and chain transfer agents ctas as well as diverse spectroscopic and electrochemical analytical techniques.
However it is challenging to incorporate these substrates in transition metal catalyzed olefin polymerization due to various side reactions such as cationic.
Facile in situ generation of vinyl ether hydrogen halide.
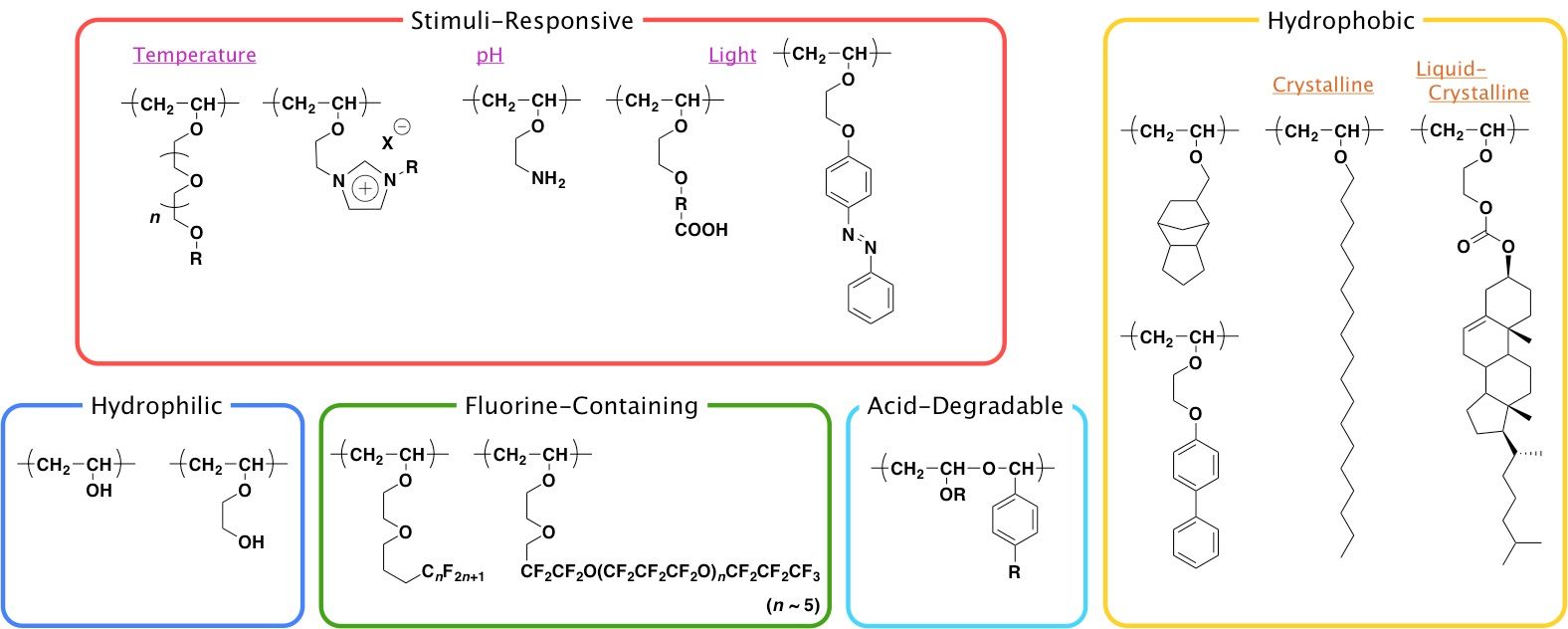
Vinyl ethers ch 2 chor r methyl ethyl isobutyl benzyl are very reactive vinyl monomers.
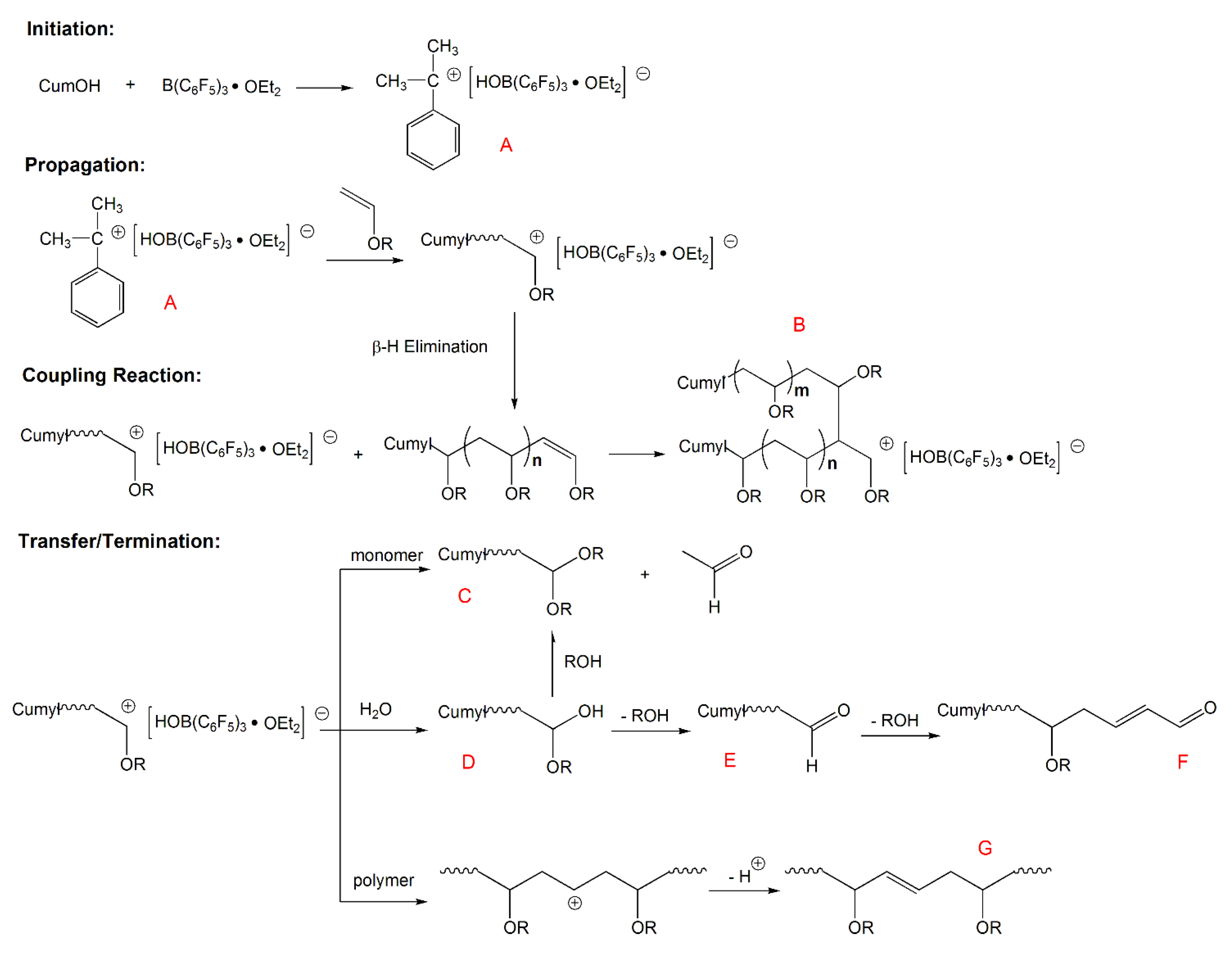
Polymerization of benzyl vinyl ether was carried out by bf 3 oet 2 and the effects of polymerization conditions on the stereoregularity of the polymer were studied by nmr analysis the polymerization at 78 c in toluene gave a highly isotactic polymer.
Living cationic ring opening polymerization.
When highly reactive cycloaliphatic epoxides are subjected to photoinitiated cationic polymerization in the presence of vinyl ethers the two polymerizations proceed in a sequential fashion with the vinyl ether.
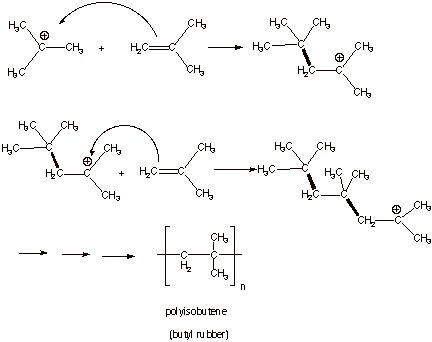
It is prone to polymerization leading to formation of polyvinyl ethers.
In all cases the rate of epoxide ring opening polymerization is accelerated whereas that of the vinyl ether is depressed.
This method however is highly sensitive to monomer structure and achieved only 76 m and 64 m in the case of n butyl bve and ethyl vinyl ether eve respectively.
Methyl vinyl ether can be made by reaction of acetylene and methanol in presence of a base.
For example the state of the art method uses a phenoxide ligated titanium complex to achieve 92 meso diads m in the polymerization of iso butyl vinyl ether ibve.
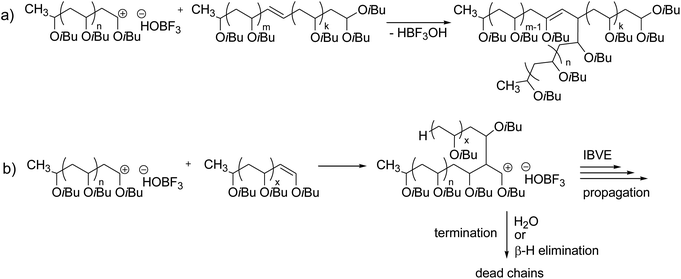
Poly vinyl ether s with a t g as high as 100 c have been obtained in the living cationic polymerization of vinyl ethers with a bulky tricyclodecane or tricyclodecene unit using hcl zncl 2 in toluene at 30 c.
Furthermore they are highly attractive monomers for the synthesis of many polymers and copolymers.
124 the fast living cationic polymerization of vinyl ethers with sncl 4 combined with etalcl 2 in the presence of an ester as an.
Kanazawa a hashizume r kanaoka s.
The alkene portion of the molecule is reactive in many ways.
Studied systems are based on i 2 hi and on zinc halides zinc chloride zinc bromide and zinc iodide.
Design of benign initiator for living cationic polymerization of vinyl ethers.
The isotacticity of the polymer was independent of the catalyst concentration but increased with a decrease in the initial monomer.
This oxidation is followed by mesolytic cleavage.
Aqueous cationic polymerizations of vinyl ethers isobutyl vinyl ether ibve 2 chloroethyl vinyl ether ceve and n butyl vinyl ether n bve were performed for the first time by a cumoh b c6f5 3 et2o initiating system in an air atmosphere.