Christensen and timothy j.
Vinyl cyclopropane synthesis.
We have developed an efficient synthesis of cyclopentanes via a ring expansion reaction of cyclopropanes embedded into a.
In this review we give an overview of their applicatio.
Enantioselective ring expansion of vinyl cyclopropanes combining four catalytic cycles for the synthesis of highly substituted spirocyclopentanes bearing up to four stereocenters cheminform 10 1002 chin 201648070 47 48 2016.
Marta meazza ramon rios cheminform abstract.
In their synthesis the construction of the chiral cyclopropane moiety relied on an enzymatic acetylation of meso diol 53 with vinyl acetate in the presence of lipase ak.
Attention was then turned to optimization of the one pot sequential allylation retro.
Unexpectedly they found that 2 s 3 r 11 s 12 r 2 r 11 s 12 r plakoside a was also identical to those reported for natural plakoside a.
Corey s synthesis of antheridiogen an 1985 elias j.
Co p1 is an effective catalyst for asymmetric cyclopropanation of various olefins with succinimidyl diazoacetate providing the desired cyclopropane succinimidyl esters in high yields and excellent diastereo and enantioselectivity.
A vinyl cyclopropane rearrangement embedded in an iridium catalyzedhydrogenborrowing reactionenabled the formation of substituted stereo defined.
The cyclopropane succinimidyl esters serve for the synthesis of optically active cyclopropyl carboxamides.
A cobalt catalyzed cyclopropanation of phenyl vinyl sulfide.
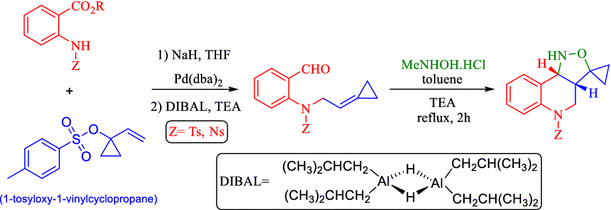
Initial experiments shown in table 1 a revealed that formation of the vinyl cyclopropane was feasible starting from the corresponding allyl ester in the presence of palladium 0.
A vinyl cyclopropane rearrangement embedded in an iridium catalyzed hydrogen borrowing reaction enabled the formation of substituted stereo defined cyclopentanes from ph methyl ketone and cyclopropyl alcohols.
Their easy opening and capacity to generate dipoles have been exploited for the synthesis of cyclopentanes with good yields and sometimes excellent stereoselectivities.
Subsequently a retro claisen condensation is utilized as a means of through space anion relay.
Corey has contributed heavily to the development of the vinylcyclopropane rearrangement as a synthetic method.
A method where an allyl alcohol is formed from a tsuji trost allylation between a vinyl epoxide and an acyl containing nucleophile is described.
Avinyl cyclopropane ring expansion and iridium catalyzed hydrogen borrowing cascade simon wbbolt choon boon cheong james r.
Vinyl cyclopropanes are amongst the most useful building blocks in organic synthesis.
In 1985 corey and his student andrew g.